Translation quality control
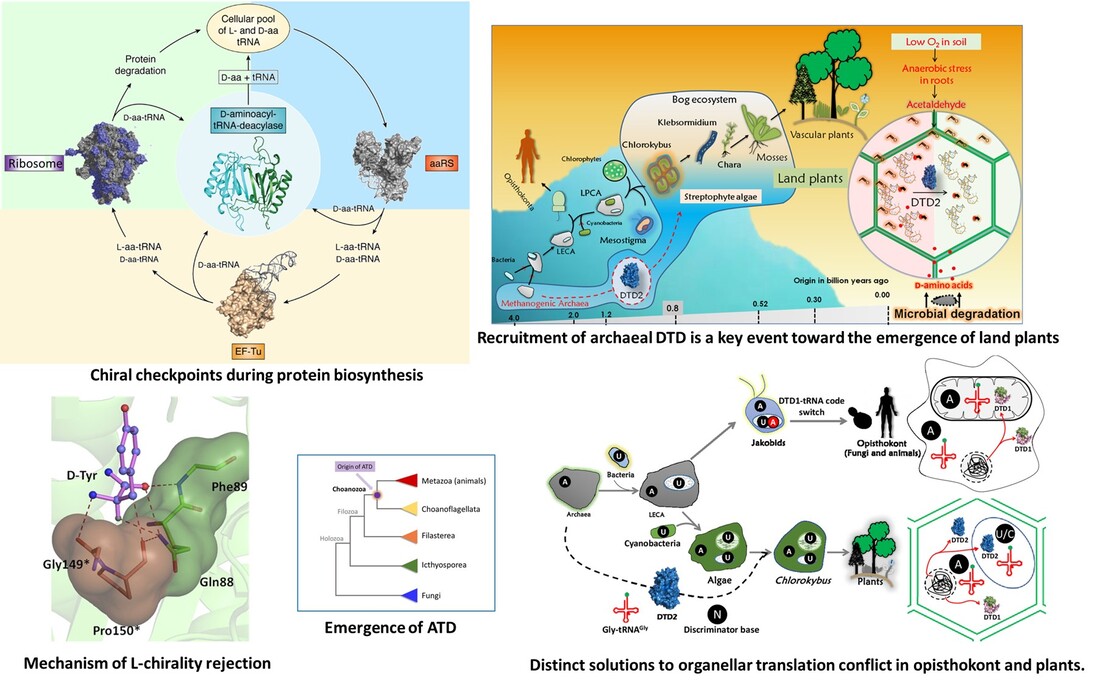
Our group is interested in understanding unique proofreading mechanisms that are operational in biological systems to maintain quality control during the translation of the genetic code. These processes are important in understanding how D-amino acids are kept away from getting incorporated during protein biosynthesis. Most of our research work is focused on understanding the mechanistic, physiological and evolutionary role of 'chiral proofreading' systems.
Our laboratory has elucidated the mechanism of D-aminoacyl-tRNA deacylase 1 (DTD1), where we have shown how an invariant ‘cross-subunit’ Gly-cisPro motif captures the chiral centre of incoming D-aminoacyl-tRNA. We have also identified a paralog of DTD1 in animals, known as Animalia-specific-tRNA Deacylase (ATD), which plays a crucial role in protecting animals from mistranslation associated with oxidative stress.
Very recently, we identified the role of archaeal-derived chiral proofreader D-aminoacyl-tRNA deacylase 2 (DTD2) in removing N-alkyl adducts formed on D-aminoacyl-tRNA by aldehydes, an anaerobic fermentation intermediate. We have shown that due to the L-chirality rejection mechanism, DTD1 also acts on glycine mischarged on tRNA(Ala), thereby preventing the misincorporation of glycine in place of alanine into proteins. We showed that, the discriminator base (N73) of tRNA protects cognate Gly-tRNA(Gly) from deacylation by DTD1. Furthermore, we went on to show how these two chiral proofreaders (DTD1 & DTD2) are involved in the optimization of cellular networks during the emergence and evolution of organelles in two major branches of eukaryotes, i.e., opisthokonta & plants.
We are currently interested in:
- Understanding the raison d'etre of DTD and its variants across life forms.
- Mechanistic underpinnings of discriminator base selection by DTD-fold.
- Mechanism of relaxed chiral selectivity of ATD.
- Elucidating the mechanism of D-chiral selectivity and adduct activity by DTD2.